✅ Built for off-grid: modular 1.2 V cells build 12/24/48 V banks
🛡️ Fail-safe by design: no BMS, non-flammable alkaline electrolyte
⚡ EMP-resilient: chemistry keeps working when electronics don’t
🌡️ Cold-ready: wide operating range
🔋 Abuse-tolerant: handles deep discharge, overcharge
🔧 Field-serviceable: performance can be renewed via electrolyte refresh
🔁 Decades of service: century-proven NiFe chemistry
♻️ Sustainable & recyclable: steel plates and alkaline electrolyte
NiFe batteries deliver decades of service without the failure modes seen in lead-acid systems.
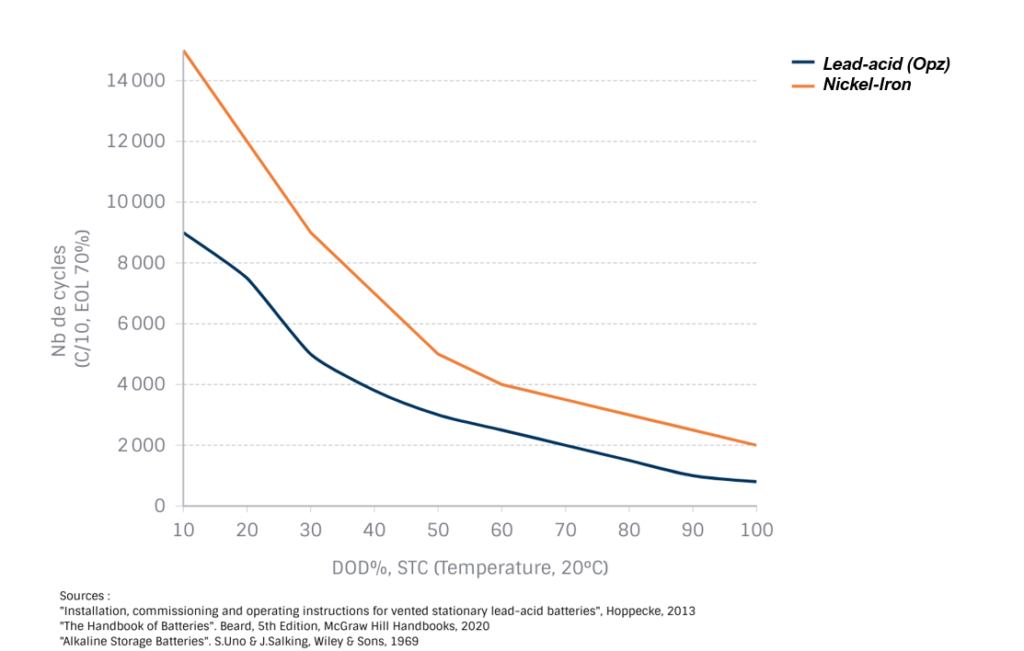
NiFe batteries are an excellent choice for remote locations where reliability and longevity are the primary factors. These batteries do not suffer from the degradation effects commonly seen in lead-acid batteries (electrolyte stratification, sulfation, electrolyte deterioration, electrode corrosion leading to internal short circuits), even in the best versions (OPZ). The average lifespan of a Nickel-Iron battery is over 20 years under normal use, with the added benefit that its electrolyte can be replaced to rejuvenate its performance. Nickel-Iron batteries have over a century of proven performance, and their behavior in the field has been extensively studied and documented in specialized literature.

Edison once claimed that his batteries were designed to last a century. While Edison, being a brilliant businessman, certainly knew how to craft a compelling sales pitch, there is documented evidence of a 1934 batch of Nickel-Iron batteries where 50% of the capacity was recovered (link).
Although they are more expensive upfront compared to the best lead-acid batteries, this is justified by the cost of raw materials (Nickel), and the investment is offset by a low total cost of ownership.
Their high tolerance to abuse (prolonged partial charge, short-circuiting, extended deep discharges, mechanical shocks, freezing) provides great peace of mind, making them particularly well-suited for the harsh conditions often encountered in off-grid setups.
Unmatched in robustness and longevity since their invention in 1901 by Thomas Edison and W. Jungner, NiFe batteries fell out of favor in the 1970s, overtaken by the more affordable lead-acid batteries, and later by lithium-ion in the 1990s. The exceptional longevity of Nickel-Iron batteries is explained by their chemical properties (see PDF study).
The structural degradation of the components (iron electrode and nickel electrode) is virtually non-existent, thanks to an alkaline electrolyte that acts as a preservative for the metals, much like in Nickel-Cadmium batteries (as opposed to acidic batteries). Historically, they were used in challenging conditions such as subways, mines, and military aviation.


For instance, some salvaged, old Nickel-Iron batteries dating back from 1930’s showed amazing capacity recovery after a couple of charging/discharging and an electrolyte remplacement. This is a testimonial to their exceptional longevity.
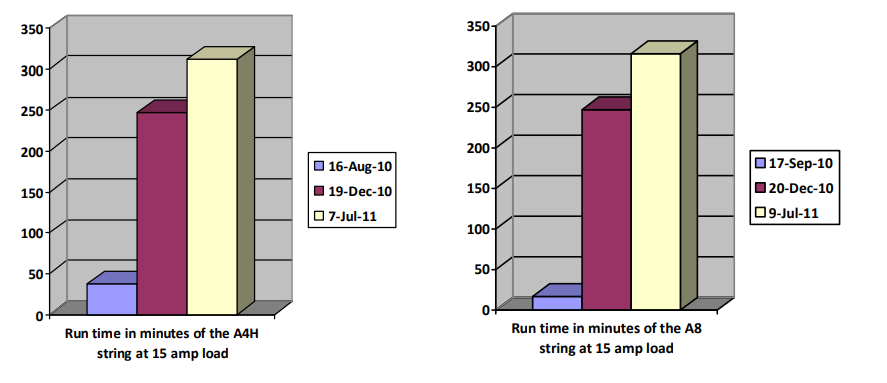
Documented old Edison batteries manufactured in 1930 that were reconditioned to a decent capacity, in 2010.
NiFe batteries use a water-based alkaline electrolyte with no flash point — eliminating lithium-style fire risks.
Nickel-Iron batteries use a non-flammable alkaline electrolyte, with no flash point, composed of an aqueous solution with approximately 25% potassium hydroxide (KOH).
Free from environmental toxicity and without harmful gas emissions, these batteries do not require electronic management systems (BMS), unlike lithium batteries, where the organic electrolyte does not provide a regulatory function in case of overcharging.
The electrolyte can be replaced by the user, using technical-grade potassium hydroxide powder and distilled or ionized water. The trade-off for this safety-oriented design is that Edison batteries have a lower efficiency (~80%) compared to lithium batteries (>95%).
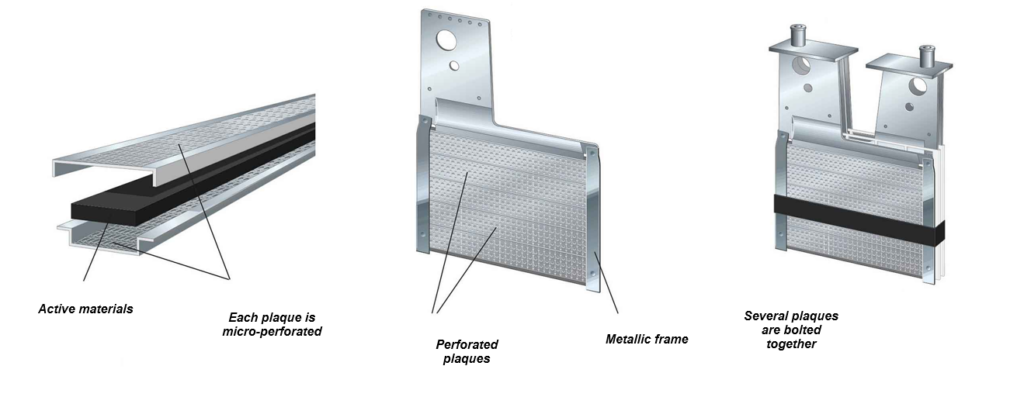
The tubular design of the battery, where the electrode materials are enclosed in finely perforated, nickel-plated tubes, provides very high mechanical resistance (to shocks).
This is precisely why Edison initially designed these batteries to be used in automobiles.
Nickel-Iron systems deliver durable, field-serviceable power you can trust when the grid can’t.
Edison (Nickel-Iron) batteries can be relied upon when the grid can’t. Built on century-proven chemistry with no onboard BMS or delicate electronics to fry (EMP-resilient by design), they’re engineered for true fail-proof operation in remote power systems. Thick steel plates and an alkaline electrolyte tolerate deep discharge, overcharge, and wide temperature swings without the thermal-runaway risks of other chemistries. Maintenance is simple—periodic distilled-water top-ups—and even after years of hard use they can be rejuvenated with an electrolyte refresh. For cabins, homesteads, telecom sites, and storm- or wildfire-prone regions, NiFe delivers durable, field-serviceable power you can count on for decades.

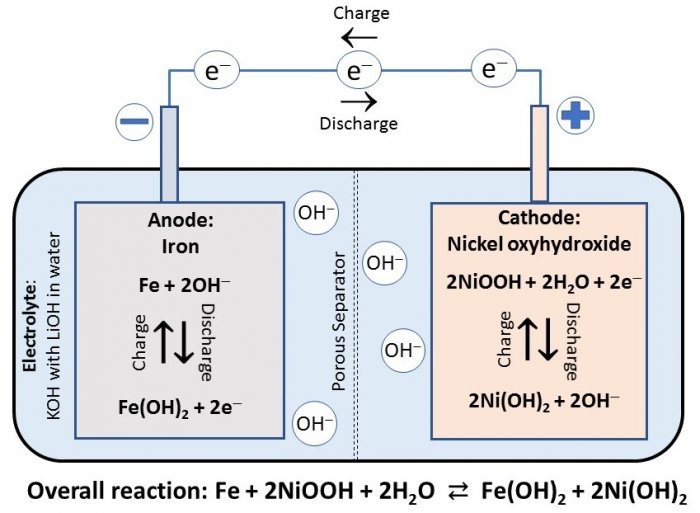
Nickel–iron stores energy through reversible solid-state redox without corrosion-driven decay.

Nickel–iron battery store energy through highly reversible, solid-state redox reactions in a strongly alkaline electrolyte (typically KOH). During discharge, the iron negative electrode is oxidized from metallic Fe to ferrous hydroxide, releasing electrons (Fe + 2 OH⁻ → Fe(OH)₂ + 2 e⁻), while the nickel positive electrode is reduced from nickel oxyhydroxide to nickel hydroxide (NiOOH + H₂O + e⁻ → Ni(OH)₂ + OH⁻). The overall reaction is a phase change between insoluble hydroxides/oxyhydroxides: Fe + NiOOH + H₂O → Fe(OH)₂ + Ni(OH)₂. Charging simply drives these steps in reverse, regenerating Fe and NiOOH. Because all active species remain as solid phases with extremely low solubility in high-pH media, the electrodes don’t “corrode” in the usual sense of dissolving or pitting; instead, they shuttle between stable oxide/hydroxide states. Alkalinity rapidly passivates iron with adherent Fe(OH)₂/Fe₃O₄ layers, and nickel operates within a potential window where transpassive dissolution is negligible. There are no intercalation hosts to crack, no dendrites to plate, and no flammable SEI to maintain. The KOH electrolyte acts as an ionic conductor and OH⁻ reservoir rather than a reactant, so it can be replaced to restore conductivity if carbonates accumulate from CO₂ ingress. This chemistry underpins NiFe’s legendary tolerance to overcharge, deep discharge, and temperature extremes, translating into decades-long service life.
Nickel-iron batteries regain performance by renewing the electrolyte — not replacing the pack.
These batteries are also uniquely “renewable” at the electrolyte level: most performance fade stems not from irreversible electrode damage but from CO₂ ingress that converts KOH to potassium carbonate (K₂CO₃), lowering pH, ionic mobility, and charge acceptance while raising internal resistance and gassing. Because the active materials remain solid and insoluble, a full drain/flush and refill with fresh KOH (optionally with a small LiOH additive for low-temperature kinetics) restores the cell’s transport properties and operating window—often recovering near-as-new efficiency. Practically, operators trigger reconditioning when they observe rising IR, sluggish charging, visible carbonate crystals, or lab checks showing significant carbonate fraction by simple two-endpoint titration (separating OH⁻ from CO₃²⁻). Rinsing plates and separators with deionized water removes carbonates, iron fines, and carbonated gel, reopening pores and wetting paths; the new electrolyte then re-establishes high conductivity without altering electrode stoichiometry. Benefits are compelling for off-grid users: service can be done in the field with basic PPE and containers, it’s low-cost and non-toxic relative to replacing a pack, and it resets calendar life without dependence on electronics or proprietary firmware. In effect, NiFe turns “end of life” into scheduled maintenance—swapping the electrolyte when carbonate accumulation becomes performance-limiting, and keeping the same rugged plates working for decades.
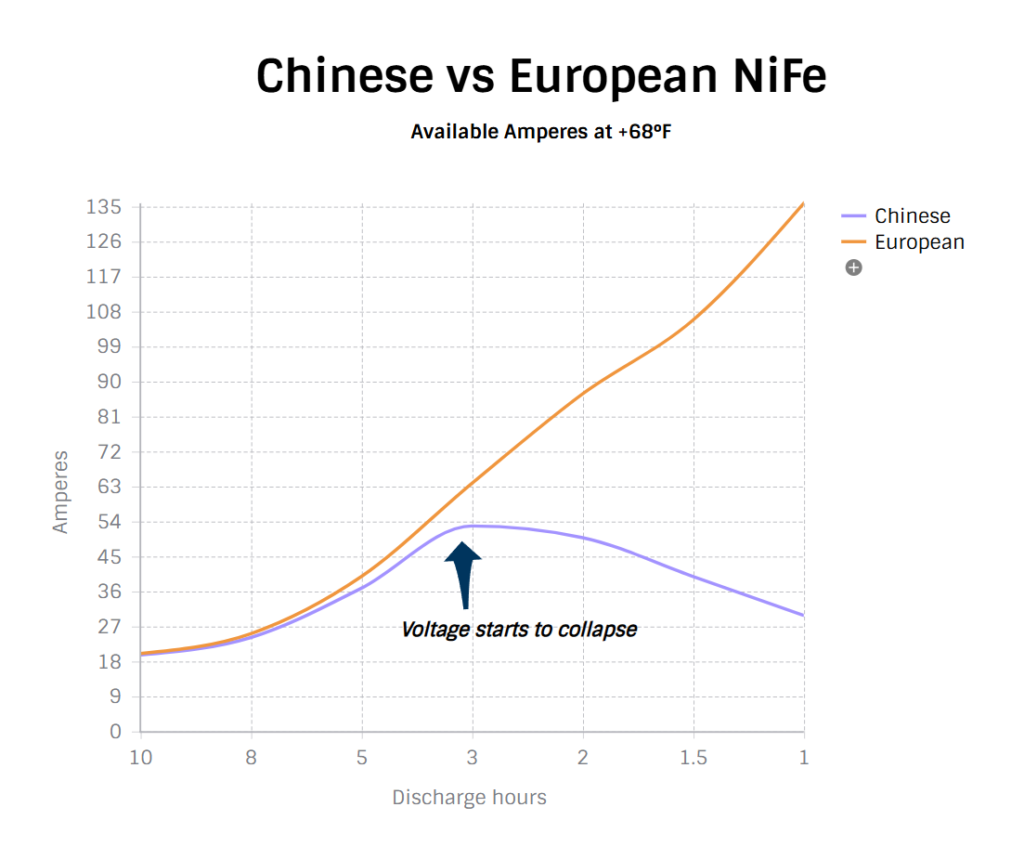
For several years, we sourced Chinese-manufactured Edison cells due to their strong availability and competitive cost-to-value ratio. More recently, however, we have transitioned to a European-based manufacturer for strategic and quality-driven reasons:
Uncompromising quality assurance — ISO-certified production with individual cell testing and traceability before dispatch.
European-sourced raw materials — steel, nickel, iron powders, and consumables sourced within the EU whenever possible, ensuring supply chain transparency and higher consistency.
Exceptional logistics and delivery speed — only 3 days from factory to our facility, enabling rapid order fulfillment before onward shipping to the US and Canada through our logistics partner.
Superior performance characteristics — flatter and more predictable discharge curves, supporting higher discharge rates (up to 1C) and tolerance to power bursts from inverters without voltage sag or collapse.


Reviews
There are no reviews yet.